Biospecimen Management Platform
Slope.IO's biospecimen management platform supports the full biospecimen lifecycle from lab kits and samples to associated metadata across pre-collection, collection, and post-collection phases. The platform is built to meet the demands of regulated environments, with a strong focus on compliance.
Shaping the foundation of a platform from the ground up.
I joined Slope at a pivotal moment, as the company began defining its next-generation biospecimen management platform, Biospecimen 360. As the sole designer on the initiative, I was responsible for leading user research, experience design, and validation efforts. The first priority was to establish clarity around the problem space. I led efforts to define the core challenges, identify key user archetypes, and align the team on who we were designing for and why. This foundational work set the direction for a more user-centered product strategy.
Project Goal: Design and deliver an efficient, intuitive biospecimen management system that supports the full operational workflow, including:
The goal was to reduce complexity, improve usability, and ensure the system supports real-world clinical workflows.
Limitations: Resource constraints led me to explore AI tools to increase efficiency and throughput. At the same time, engineering capacity and readiness were still maturing, which impacted overall delivery timelines.
Understanding the problem space end-to-end.
I started with understanding the problem space end-to-end. I began by synthesizing product feedback and deeply analyzing existing workflows to identify structural gaps in the experience. It became clear that the product was not aligned with how clinical teams actually operated in their environments.
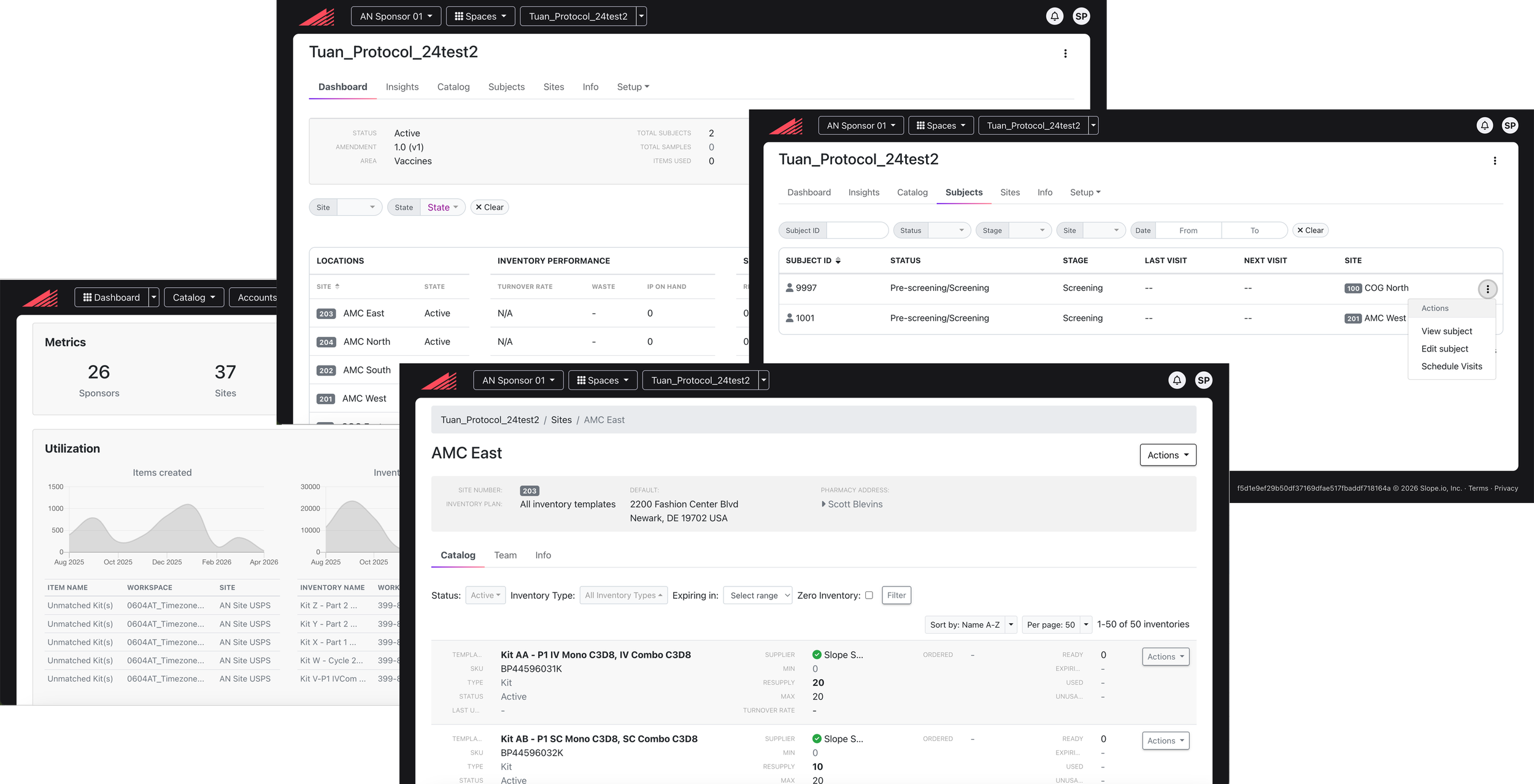
The existing product — analyzing current workflows to identify structural gaps in the experience.

Photos from my own site visits — shadowing clinicians and lab technicians to observe biospecimen workflows firsthand.
To bridge this gap, I conducted user research including interviews and shadowing clinicians in their physical workspaces. This allowed me to observe firsthand how biospecimen workflows unfold in real-world settings. Through ethnographic research, I developed a nuanced understanding of the different roles involved — those directly interacting with the system and those who required visibility for oversight, compliance, and coordination.
These insights shaped a strategic shift from feature-based thinking to a systems-driven approach. I established a measurement framework to define success, grounded in usability, adoption, and compliance outcomes. From there, I defined core user archetypes, mapped end-to-end journeys, and outlined the critical workflows and reporting structures required to support regulated environments.
This work laid the foundation for a more intuitive, scalable, and compliance-ready platform — one that aligns with real-world clinical operations and supports both execution and oversight across the biospecimen lifecycle.
Exploring the problem space with the full team.
We then conducted a design workshop where we explored the problem space deeply, and defined the problems we wanted to focus on.
We then broke the team into groups to brainstorm features within each of these problem spaces and possible solutions.
We had the engineering team help us understand technical limitations and sales help us understand all the compliance issues we will need to support.





Exploring versatile, intuitive sample and subject interlaced timepoint flows.
Building alignment before building the product.
Equipped with research and clarity, I started with building a slope value model for the teams to align on. Once we were in alignment on the platform we were building and the archetypes we were building for, we cleaned up the personas.
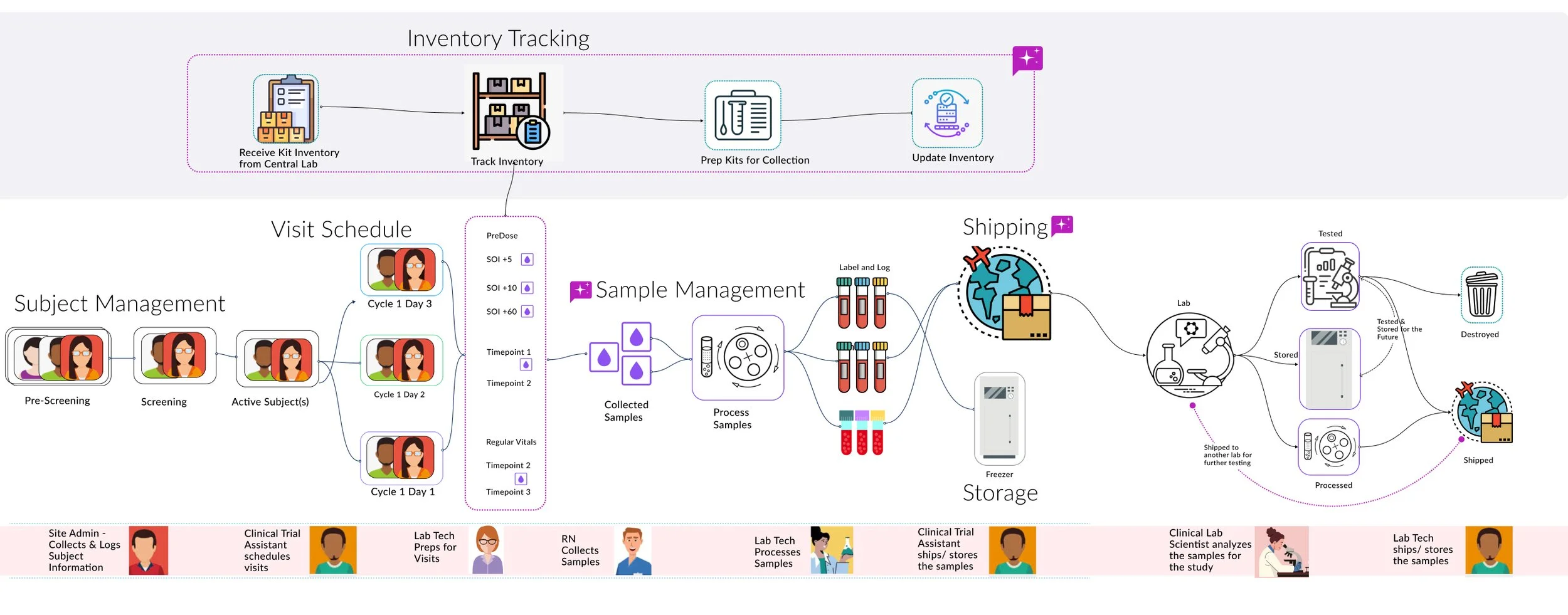
Platform model — mapping the full biospecimen lifecycle across inventory tracking, visit scheduling, sample management, shipping, and storage.

Sponsor and CRO Coordinator archetypes — defining oversight and operational roles.

Lab Technician archetype — grounding collection and processing workflows in real user needs.
From archetypes to shipped product.
I established the product vision, which was translated into features, user stories, and sprint-level designs in partnership with product management. Biospecimen 360 has received positive validation in testing, with adoption across 200+ clinical sites and $10M+ in contracts secured with large pharmaceutical companies.
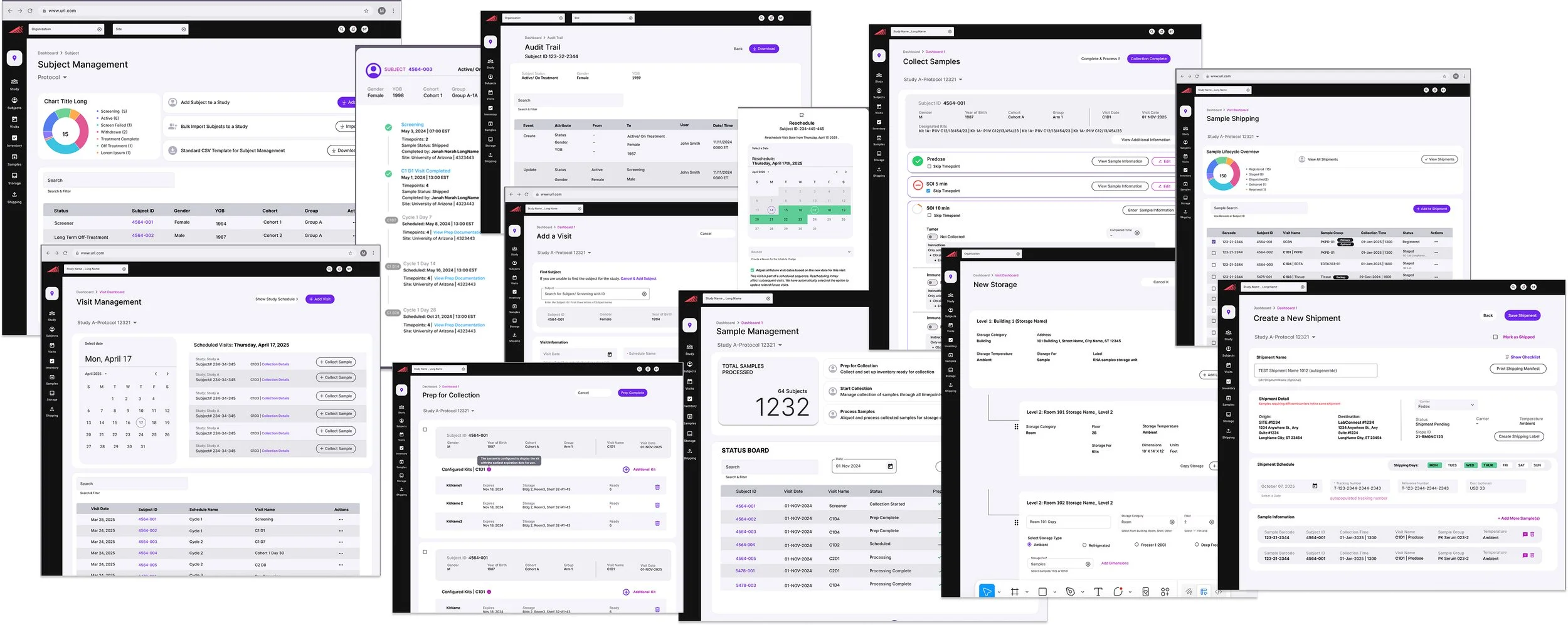
Biospecimen 360 — final designs covering Subject Management, Visit Scheduling, Sample Collection, Storage, and Shipment workflows.